Magnesia-carbon refractories emerged in the 1970s and are now widely used in modern steel metallurgy, such as in the linings of various electric arc furnaces, ladles, and steelmaking refining furnaces (BOF, RH). Magnesia-carbon bricks are alkaline refractory materials made from magnesia and graphite using various binders. Graphite, as a crucial component of magnesia-carbon refractories, has a vital influence on their performance. Due to graphite’s high thermal conductivity, low coefficient of thermal expansion, and non-wetting properties with molten slag,
adding graphite significantly improves the thermal shock resistance and slag erosion resistance of magnesia-carbon refractories. However, graphite is prone to oxidation, leading to an increase in the porosity of magnesia-carbon bricks, which in turn reduces strength and erosion resistance, causing material peeling and structural damage. Therefore, research on the oxidation resistance of magnesia-carbon refractories is of great practical significance. Graphite oxidation can be divided into direct oxidation and indirect oxidation. Direct oxidation mainly refers to the reaction between graphite and oxygen (Equation 1); while indirect oxidation mainly refers to the reaction between graphite and magnesium oxide at high temperatures (Equation 2). It is generally believed that direct oxidation is the primary process when the temperature is below 1400℃, while indirect oxidation plays a major role when the temperature is above 1400℃.
2C(s) + O2(g) → 2CO(g) (1)
MgO(s) + C(s) → 2Mg(g) + CO(g) (2)
This paper focuses on the oxidation resistance of magnesia-carbon bricks with different graphite contents. By measuring the bulk density and porosity of magnesia-carbon brick samples before and after carbonization, as well as the thickness of the decarburized layer after oxidation, the oxidation rate of magnesia-carbon bricks with different graphite contents was calculated using an oxidation model.
Experimental Section
1.1 Raw Materials and Experimental Scheme
The magnesia-carbon bricks required for this experiment were prepared using fused magnesia with a purity of 98.5% and natural flake graphite with a purity of 99.3% as raw materials and resin as a binder.
The main preparation process of magnesia-carbon brick samples is as follows: First, resin is uniformly adhered to the surface of coarse magnesia particles, then fine magnesia powder and graphite are added sequentially, and the mixture is stirred evenly using a ball mill until no agglomerates are present. The uniformly mixed magnesia-carbon brick raw materials are then pressed into cylindrical samples with a diameter of φ50mm × 40mm using a pressing tester. The molding pressure is 5MPa, and the holding pressure is maintained for 10min.
1.2 Experimental Procedure and Performance Testing
The corundum crucible containing the magnesia-carbon brick sample was placed in a tube-type resistance furnace for carbonization testing. The furnace was heated to 900℃ at a rate of 5℃ and held for 1 hour. Argon gas protection was used throughout the test to prevent oxidation of the magnesia-carbon brick sample. The bulk density (Equation 3) and porosity (Equation 4) of the magnesia-carbon brick before and after carbonization were measured using the Archimedes method. The prepared magnesia-carbon brick was cut into 30×30×30mm cubic samples for oxidation testing. The oxidation temperatures were 1000℃ and 1400℃, and the holding time was 4 hours. After the oxidized samples cooled, the oxidized magnesia-carbon brick was cut along its cross-section, and the oxide layer thickness was measured.
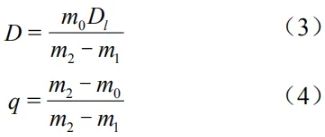
In the formula: D—bulk density;
m0—mass of the dry sample in air;
m1—mass of the saturated sample in the liquid medium;
m2—mass of the saturated sample in air;
Q—apparent porosity;
D1—density of the liquid medium.
Results and Analysis
Figure 2 shows the bulk density and porosity of magnesia-carbon brick samples with different graphite contents before and after carbonization. With increasing graphite content, the bulk density of the magnesia-carbon bricks before carbonization decreased while the porosity increased. For example, when the graphite content increased from 5% to 20%, the bulk density of the magnesia-carbon brick samples decreased from 3.08 g·cm⁻³ to 2.93 g·cm⁻³, while the porosity increased from 2.19% to 3.31%. The decrease in bulk density of the magnesia-carbon brick samples is due to both the increase in porosity and the fact that graphite itself has a lower density than magnesium oxide. After carbonization at 900℃, the bulk density of the magnesia-carbon bricks decreased compared to before carbonization, while the porosity significantly increased to over 10%.
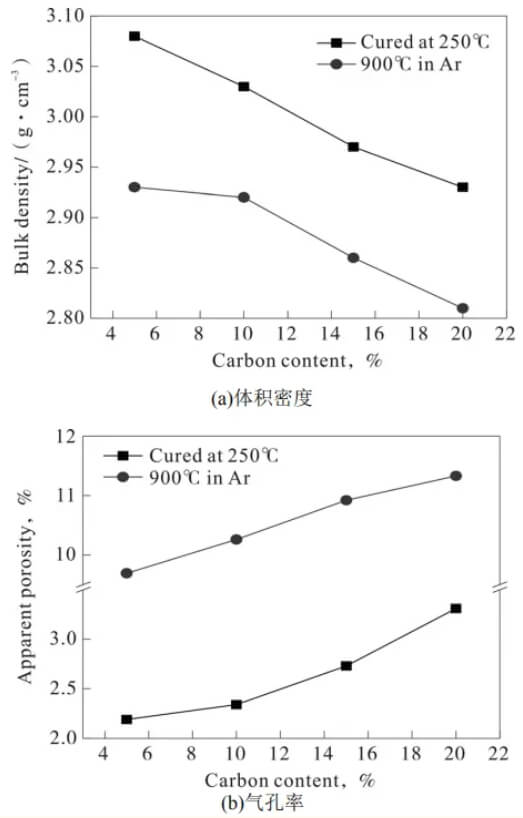
Figure 3 shows the decarburized layer thickness of magnesia-carbon brick samples after oxidation at 1000℃ and 1400℃ for 4 hours. As can be seen from the figure, the decarburized layer of the magnesia-carbon brick sample oxidized at 1400℃ is significantly thicker than that oxidized at 1000℃, indicating that the increased temperature increases the oxidation rate and makes the oxidation more severe. Furthermore, the decarburized layer thickness of the magnesia-carbon brick samples decreases with increasing graphite content, but when the graphite content increases to 10% or higher, the change in graphite content has little effect on the decarburized layer.
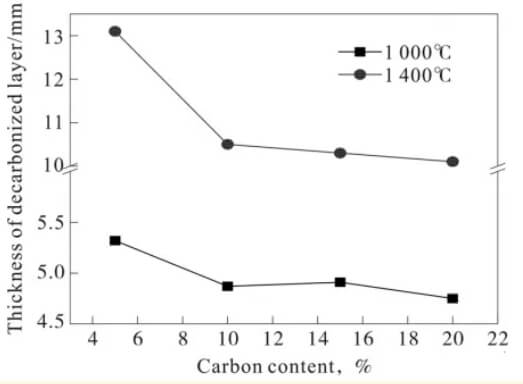
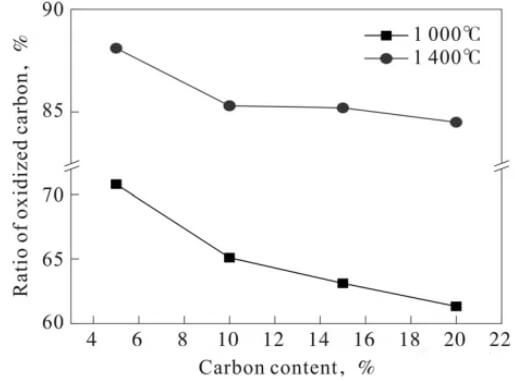
Figure 4 shows the graphite oxidation rate of magnesia-carbon brick samples with different graphite contents after oxidation at 1000℃ and 1400℃ for 4 hours. As can be seen from Figure 4, the trend of graphite oxidation rate is basically consistent with the trend of decarburized layer change. That is, the graphite oxidation rate decreases with increasing graphite content. Furthermore, the graphite oxidation rate of the magnesia-carbon brick sample oxidized at 1400℃ is significantly higher than that of the sample oxidized at 1000℃.
The oxidation rate of the magnesia-carbon brick sample during the high-temperature oxidation process can be expressed by equation (5).
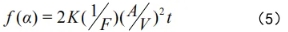
In the formula: a—oxidation rate;
K—reaction rate constant;
F—shape factor of the magnesia-carbon brick sample;
A—sample surface area;
V—sample volume;
t—oxidation time.
Assuming that the magnesia-carbon brick sample is unidirectional, the oxidation rate can be written in the form of equation (6).
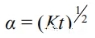
Reaction rate:
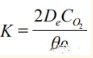
In the formula: De—Effective diffusion coefficient of oxygen in the pores of the refractory material;
CO2—Oxygen concentration outside the refractory material;
—Volume fraction of graphite in the magnesia-carbon brick;
—Density fraction of graphite in the magnesia-carbon brick.
Effective diffusion coefficient:
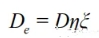
In the formula: D—diffusion coefficient of oxygen within the pores of magnesia-carbon bricks;
—porosity of the decarburized layer of the magnesia-carbon bricks;
ξ—maze coefficient.
And the maze coefficient:
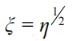
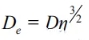
Furthermore, the porosity of the decarburized layer (η) is the sum of the porosity (0) of the carbonized magnesia-carbon brick and the porosity (C) of the oxidized magnesia-carbon brick.
η = η0 + ηC (11)
The porosity of the oxidized magnesia-carbon brick is equal to the volume fraction of graphite in the magnesia-carbon brick, and is also equal to equation (12).
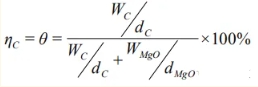
Wherein: WC and WMgO represent the contents of graphite and magnesium oxide in the magnesia-carbon brick, respectively;
dc and dMgO represent the densities of graphite and magnesium oxide in the magnesia-carbon brick.
Substituting equations (7) to (12) into equation (6) yields the oxidation rate of the magnesia-carbon brick sample, as shown in Figure 5. Figure 5 shows that the oxidation rate of the magnesia-carbon brick sample gradually decreases with increasing graphite content. When the graphite content is 5%, the oxidation rate of the magnesia-carbon brick sample reaches over 20%; however, when the graphite content increases to 20%, the oxidation rate decreases to approximately 10%. Simultaneously, when the graphite content reaches over 10%, the change in the oxidation rate of the magnesia-carbon brick sample is relatively small. The trend of graphite oxidation rate calculated by the model is largely consistent with the actual measured trend of decarburized layer thickness.
Conclusion
(1) The bulk density of the magnesia-carbon brick samples decreased with increasing graphite content, while the porosity increased. After carbonization at 900℃, the bulk density of the magnesia-carbon brick samples decreased slightly, while the porosity increased significantly.
(2) The higher the graphite content in the magnesia-carbon brick samples, the thinner the decarburized layer and the lower the graphite oxidation rate. Furthermore, the magnesia-carbon brick samples oxidized at 1400℃ had lower decarburized layer thickness and graphite oxidation rate than those oxidized at 1000℃.
(3) The oxidation rate of the magnesia-carbon brick samples calculated using the oxidation model showed a trend consistent with the actual measured trend of the decarburized layer thickness. That is, the oxidation rate decreased with increasing graphite content, and the decrease was most significant at lower graphite contents.
